Improving IV Solution Manufacturing Efficiency: Key Technologies for 0.3% Filling Accuracy?
You are running a busy pharmaceutical plant, and every drop of liquid counts. You worry about waste, you worry about safety, and you worry about meeting strict regulations. If your IV bags are not filled perfectly, you lose money and risk your reputation.
Achieving 0.3% filling accuracy is the key to solving these problems, and it requires specific technology like mass flow meters and advanced PLC controls found in modern SRD and SRDS series machines. This level of precision ensures patient safety and maximizes your production profits.

IV bag filling machine accuracy
Many factory managers think that a little variation in filling volume is acceptable, but I have seen how small errors destroy profit margins over time. Let’s look at why this specific number—0.3%—is the target you must hit.
1.Why is 0.3% accuracy the "Gold Standard" for 2000ml IV bags in modern pharma plants?
You might ask yourself if such high precision is really necessary for a simple saline or glucose solution. The answer is a definite yes because the cost of errors is much higher than you think.
0.3% accuracy is the gold standard because it represents the perfect balance between patient safety standards and strict cost control for raw materials. It ensures that every single bag leaving your facility is compliant without giving away free product.

Standard 2000ml IV bag production
When we talk about filling 2000ml bags, a 1% error means a variance of 20ml. If you produce 50,000 bags a day, that 1% overfill wastes 1,000 liters of product daily. Over a year, that is a massive financial loss. However, the issue goes deeper than just money.
In the pharmaceutical industry, "close enough" is not good enough. Regulatory bodies like the FDA or EMA have strict guidelines on dosage. If a bag is underfilled, the patient does not get the required treatment. If it is overfilled, you are wasting expensive Water for Injection (WFI) and active pharmaceutical ingredients (API).
We must also consider the downstream effects. If a bag is overfilled, it might be under too much pressure during sterilization or transport, leading to bursts. If it is underfilled, the concentration of the solution might be questioned.
Here is a breakdown of why 0.3% is the target:
| Factor | Impact of Low Accuracy (>1%) | Impact of High Accuracy (0.3%) |
| Raw Material Cost | High waste of API and WFI | Significant savings on expensive ingredients |
| Patient Safety | Risk of incorrect dosage | Guaranteed therapeutic consistency |
| Packaging Integrity | Overfilled bags may burst during transport | Stable internal pressure prevents leaks |
| Regulatory Audit | High risk of non-compliance fines | Easy validation and GMP compliance |
By aiming for 0.3%, we help our clients secure their bottom line and their reputation.
Now that we know why accuracy matters, we need to understand the technology that beats the old methods.
2.How does mass flow metering compare to traditional time-pressure filling systems?
You may still be using older machines that rely on time-pressure filling, but you have likely noticed that accuracy drifts as the tank level changes.
Mass flow metering is superior because it measures the actual weight of the liquid in real-time, regardless of temperature or viscosity changes, whereas time-pressure systems struggle when tank pressure fluctuates.
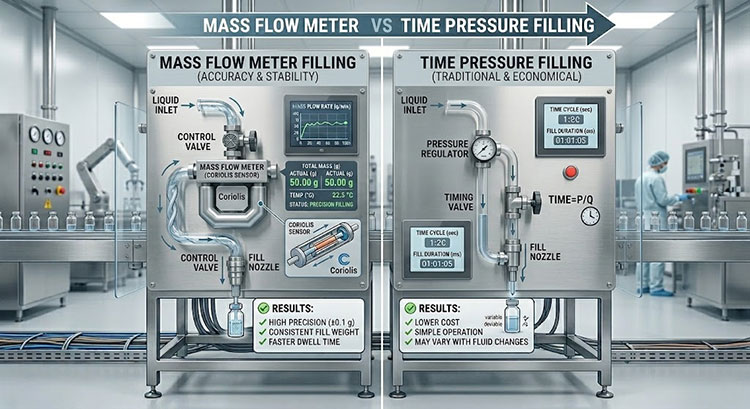
Mass flow meter vs time pressure filling
I remember visiting a client who was frustrated because his filling volume changed every time the main supply tank got half empty. This is the weakness of time-pressure systems. They open a valve for a set time, assuming the flow rate is constant. But gravity changes that flow rate as the tank empties.
Mass flow meters (Coriolis meters) work differently. They do not care about gravity or pressure. They measure the mass of the fluid passing through the tube. This means the temperature of your liquid can change, or the viscosity can shift slightly, and the machine will still deliver exactly 2000ml (or your target weight) within that 0.3% margin.
Consider these critical differences:
Viscosity Handling: In older systems, if your sugar solution cools down and gets thicker, it flows slower. The timer stays the same, so you underfill. A mass flow meter sees the flow is slower and keeps the valve open longer to compensate automatically.
Calibration: Time-pressure systems need constant recalibration throughout the day. Mass flow systems are robust and hold their calibration for much longer periods.
Maintenance:Mass flow meters have no moving parts inside the flow path. This means less wear and tear compared to piston pumps that have seals which can leak and contaminate the product.
At AIPAK ENGINEERING, we moved away from simple time-pressure systems for high-value IV lines because our customers demanded consistency. We realized that the initial investment in mass flow technology pays for itself by eliminating the "drift" that causes rejected batches.
This brings us to the brain of the operation—how the machine controls these advanced sensors.
3.How do SRD and SRDS series machines use PLC and mass flow technology to monitor filling parameters in real-time?
Having a good sensor is useless if the machine's computer cannot react fast enough to the data it provides.
The SRD and SRDS series machines use a high-speed PLC to create a closed-loop feedback system, where the mass flow meter sends data to the controller, which immediately adjusts the filling valves to correct any deviation.

AIPAK SRD machine PLC screen
In the past, machines were "open loop." You told the machine what to do, and you hoped it did it. Today, with our SRD and SRDS series, the machine is constantly "talking" to itself.
Here is how the process works inside our equipment:
Start:The PLC opens the filling nozzle.
Measure: The mass flow meter counts the fluid mass instantly.
Predict:As the weight approaches the target (e.g., 1980ml out of 2000ml), the PLC calculates exactly when to close the valve to account for the "flight" of the liquid (the liquid still in the air after the valve closes).
Act:The valve shuts precisely at the calculated moment.
Verify: The meter confirms the final weight. If it is off by even 0.1%, the PLC adjusts the timing for the *next* bag automatically.
This intelligent adjustment is vital. It means the machine gets smarter as it runs.
We also use the PLC for data integrity. In modern pharmaceutical manufacturing (GMP), if you didn't document it, it didn't happen. Our systems record the fill volume of every single bag. You can download this data to prove to auditors that your batch was perfect.
Real-time Monitoring: Operators can see the flow rate on the touch screen.
Alarm Systems:** If a nozzle gets blocked or a valve fails, the system stops immediately and tells you exactly where the problem is.
User Management:The PLC ensures only authorized engineers can change the critical filling parameters.
This technology ensures the liquid goes in correctly, but we must also ensure the environment remains sterile.
4.Can automatic CIP/SIP systems integrated into the filling line prevent cross-contamination?
Speed and accuracy are great, but if your machine is difficult to clean, you will lose hours of production time and risk bacterial growth.
Integrated CIP (Clean-in-Place) and SIP (Sterilize-in-Place) systems prevent cross-contamination by automatically washing and sterilizing the filling lines without the need for operators to disassemble the machine.

Automatic CIP SIP cleaning process
I often tell factory owners that the most dangerous time for a product is during the changeover or cleaning. Human error is the biggest risk. If an operator forgets to sanitize a gasket, the whole batch is ruined.
Our automated CIP/SIP systems remove the human element from the cleaning cycle.
Here is why this is critical for IV solution manufacturing:
Consistency:The machine follows a strict recipe. It uses the exact temperature, chemical concentration, and time every single wash. It never gets "tired" or "lazy."
Safety:Operators do not need to handle strong acids or bases used for cleaning. The system is fully enclosed.
Sterility Assurance: The SIP cycle uses high-temperature steam to kill all microorganisms. The PLC monitors the temperature at the coolest point of the system to ensure 100% sterilization.
We design the piping in our SRD series with a specific slope and no "dead legs" (areas where water can sit stagnant). This sanitary design is essential. You cannot achieve 0.3% accuracy if your nozzles are clogged with residue from the previous batch. The cleaning process ensures the flow path is perfectly clear, which aids in maintaining that high filling precision.
Cleaning protects the product, but mechanical stability protects the process. Let's look at how we handle the bags.
5.What is the relationship between bag width (120/130/135 type) and filling stability at high speeds?
You might not think the physical size of the bag matters much for filling accuracy, but at high speeds, the bag's behavior is critical.
The bag width dictates how the liquid settles during high-speed filling; matching the correct nozzle speed and gripper width to the bag size (120mm, 130mm, or 135mm) prevents splashing and ensures the bag does not wobble, which causes filling errors.
Different IV bag widths 120 130 135
When you fill a flexible IV bag quickly, the liquid hits the bottom and creates turbulence. If the bag is too narrow for the flow rate, the liquid can splash back up and touch the sealing area. This creates a bad seal (leaks) and can fool the sensors.
We encounter three main standard widths:
120mm:Often used for smaller volumes or compact packing.
130mm: A standard middle ground.
135mm:Used for larger volumes like 2000ml to reduce the height of the bag.
Here is how we adjust for these differences using critical thinking and engineering:
The "Ballooning" Effect: As liquid enters, the bag expands. A wider bag (135mm) expands differently than a narrow one. Our grippers must hold the bag firm enough so it doesn't drop, but loose enough to allow this expansion. If the gripper is too tight, the internal pressure rises artificially, fighting the filling pump.
Nozzle Design: For a narrower 120mm bag, we might use a thinner nozzle tip to allow air to escape around it. If air cannot escape (air binding), the fill will stop prematurely.
Sloshing Control: Wider bags allow the liquid to spread out fast, reducing the vertical splash. This usually allows for higher filling speeds. However, they require a wider transport footprint on the machine.
Our SRDS machines are designed with adjustable guides. When we help a client set up, we do not just look at the volume; we analyze the bag geometry. We program the "filling curve"—starting slow to open the bag, filling fast for the bulk, and slowing down at the end to prevent foam. This curve must be customized based on the bag width to maintain that 0.3% accuracy.
But even with perfect filling, a leak can ruin everything.
6.How does leakage detection technology ensure zero defects before final packaging?
You can fill a bag with 0.3% accuracy, but if it has a microscopic hole, that precision is wasted because the product is non-sterile.
Leakage detection technology, such as high-voltage detection or pressure decay testing, ensures zero defects by identifying and automatically rejecting any bag that is not perfectly sealed before it reaches the boxing stage.

High voltage leak detection for IV bags
I have seen production lines running at high speed where a tiny pinhole in one bag leaked saline onto 50 other bags in the collection bin. The workers had to clean and dry all of them manually. It was a nightmare.
Automated leak detection is the final gatekeeper.
High Voltage Leak Detection (HVLD): This is very effective for IV bags. We pass a high voltage current through the bag. Plastic is an insulator, so electricity shouldn't pass. But if there is a pinhole, the liquid inside conducts the electricity. The machine detects the spark and rejects the bag instantly.
Pressure Decay: We squeeze the bag between two plates and measure the resistance. If the bag deflates slightly, there is a leak.
Integrating this into the line is part of the "efficiency" in the title. If you catch the leak immediately after filling and sealing, you don't waste time labeling or cartoning a bad product. It also provides feedback. If the leak detector starts rejecting bag #4 repeatedly, the PLC tells us to check Sealing Station #4. It closes the loop on quality control.
Technology is great, but machines need humans to set them up correctly.
7.How does AIPAK’s technical service team help clients achieve this precision through onsite calibration and validation?
Buying the best machine in the world is not enough if it is not installed and calibrated by someone who understands the process deeply.
AIPAK ENGINEERING’s technical service team ensures you achieve precision by performing rigorous onsite Site Acceptance Tests (SAT), calibrating the mass flow meters to your specific liquid properties, and training your staff on GMP-compliant operation.**

Engineer calibrating pharma machine
I recall a project where a client bought a high-end machine but tried to install it themselves to save money. They couldn't get the accuracy below 1%. When our team arrived, we found that the supply pump was creating vibrations that confused the sensors. We fixed the mounting, adjusted the PID parameters in the PLC, and within two hours, they were hitting 0.25%.
Our support process is comprehensive:
IQ/OQ/PQ Validation:We provide the documentation required by the FDA/GMP. We prove the machine is installed correctly (IQ), operates correctly (OQ), and performs consistently (PQ).
Liquid-Specific Tuning:Saline behaves differently than a glucose or amino acid solution. We run your actual product through the machine and fine-tune the flow parameters onsite.
Training:We teach your operators not just which button to press, but *why*. When they understand that air bubbles in the supply line affect accuracy, they become better operators.
We do not just sell equipment; we sell a production capability. Our goal is to make sure that when we leave your factory, your line is running smoothly, efficiently, and accurately.
Conclusion
To achieve 0.3% filling accuracy in IV solution manufacturing, you need a combination of mass flow technology, intelligent PLC control, and stable mechanical handling. At AIPAK ENGINEERING, we combine these technologies with expert support to ensure your production is safe, compliant, and profitable.
Don't forget to share this post!
CONTACT US
Tell us your raw material and project budget to get quotations within 24 hours.
WhatsApp Us: +86 181 7101 8586
 Tell us your material or budget, we'll reply you ASAP within 24 hours
Tell us your material or budget, we'll reply you ASAP within 24 hours

