Designing a Seamless IV Fluid Production Line:From Engineering Design to Project Management
Struggling to set up your IV fluid production line? Bad layouts and wrong machines waste money and delay your launch. I will show you how to build a perfect, compliant factory.
A seamless IV fluid production line requires precise engineering design, the right equipment, and strict project management. You must integrate cleanroom layouts, water systems, and automated filling machines to meet GMP standards and ensure safe, high-volume manufacturing.

Seamless IV Fluid Production Line
You do not want to fail a GMP audit after spending millions of dollars. Let us look closely at how to build a perfect IV fluid factory step by step.
1.How does the initial engineering design phase prevent costly mistakes in liquid drug formulation and cleanroom spatial layout?
Planning a cleanroom without a good design leads to total disaster. You might face cross-contamination or wasted floor space. A good engineering design fixes these problems early.
The initial engineering design stops costly mistakes by mapping out material flow and human traffic. It ensures your liquid drug formulation area and cleanroom layout meet strict GMP rules before you buy any machines.

Cleanroom Spatial Layout Design
The Importance of Good Layout
I remember a client who built a room too small for their mixing tanks. They had to break a wall. This mistake cost them two months of time. We always start with a clear drawing. We separate the dirty areas from the clean areas. We plan the path for workers.
We plan the path for materials. This strategy stops people from bringing dirt into the cleanroom. Our engineering team looks at your building first. We place the liquid drug formulation tanks in the best spot. We make sure the pipes are short. Short pipes are very easy to clean.
You must think about the future. You need space if you want to add a second line later. We draw a 3D model for you. You can see the whole room before you spend money. A good spatial layout saves energy too. Your air conditioning system works less when the room size is right. We check all local building rules. We make sure your factory is safe and legal.
| Design Feature | Benefit for Production |
| Short Pipe Routes | Easy to clean and reduces drug waste |
| Separate Traffic Paths | Prevents human dirt from entering clean areas |
| Future Space Planning | Allows easy expansion without stopping production |
| 3D Modeling | Finds space problems before construction starts |
2.What are the key differences in designing a line for Soft Bag IV solutions versus Blow-Fill-Seal (BFS) technology?
Choosing the wrong packaging technology hurts your profit margin. Soft bags and BFS systems need totally different factory setups. You must pick the right one for your market.
Soft bag lines use pre-made bags and need separate film feeding, printing, and filling steps. BFS technology forms, fills, and seals the container in one continuous motion inside a sterile machine.
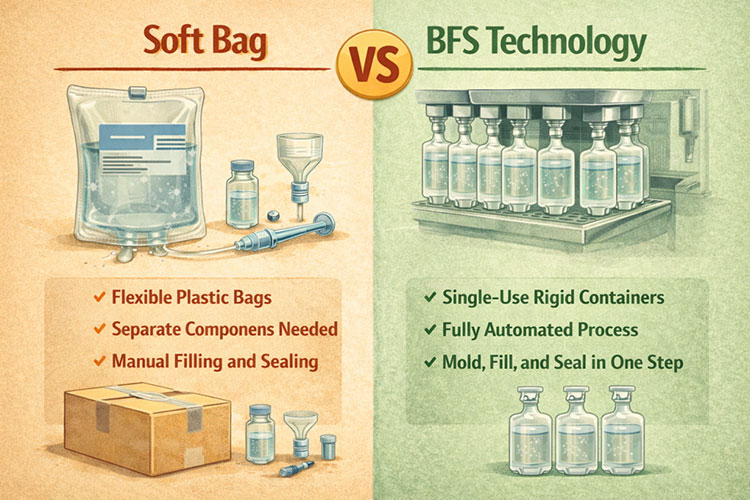
Soft Bag vs BFS Technology
Choosing Your Packaging Method
I talk to many customers about this choice. Soft bags are very popular in big hospitals. They are easy to store in small spaces. But soft bag lines need more factory floor space. You need a machine to feed the plastic film.
You need a machine to print words on the bag. BFS stands for Blow-Fill-Seal. This machine does everything in one closed box. It makes the plastic bottle from raw plastic. It fills the liquid drug. It seals the top. It does this in seconds.
BFS is very safe. The liquid never touches the outside air. But BFS machines cost more money at the start. You must look at your product type. Soft film is better if you make big IV bags. BFS is great if you make small eye drops or small IV bottles. We help you look at the cost per unit. We help you make the most money from your factory.
| Feature | Soft Bag IV Line | BFS Technology |
| Factory Space | Needs more space for many machines | Needs less space (all-in-one machine) |
| Initial Cost | Lower initial equipment cost | Higher initial equipment cost |
| Product Size | Best for large bags (100ml to 1000ml) | Best for small bottles and vials |
| Sterility Risk | Low risk | Extremely low risk (closed system) |
3.How do we determine the optimal automation level (Manual vs. Semi-automatic vs. Full-automatic) based on the local environment and production needs?
Paying for too much automation kills your initial budget. But too little automation slows down your daily production. You need the perfect balance for your location.
We determine the automation level by looking at your local labor costs, target output, and factory size. Full automation suits high-volume markets, while semi-automatic lines work best for lower budgets and cheaper labor.
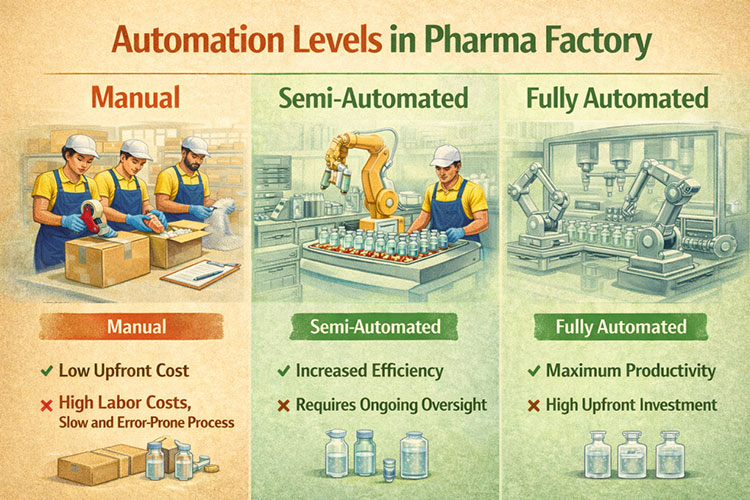
Automation Levels in Pharma Factory
Finding the Right Speed
I always ask my clients about their local workers. You need full automation if you build a factory in a place with high wages. Machines do not ask for sick days. A fully automatic line takes the empty bags, fills them, and packs them in boxes. You only need one or two people to watch the computer screens. But semi-automatic is a smart choice if your local wages are low. You spend less money on machines. Workers can load the empty bags by hand.
You still get good product quality. You just save money on the initial setup. You must also think about your daily target. Human hands are too slow if you need 100,000 bags a day. You must use full automation for big numbers. We look at your budget carefully. We look at your sales target. We find the perfect balance for your factory. We make sure you do not waste money.
| Automation Level | Best For | Labor Requirement |
| Manual | Very small test batches or research | High |
| Semi-automatic | Low labor cost regions, medium output | Medium |
| Full-automatic | High labor cost regions, huge output | Very Low |
4.What specific machinery components are essential for a seamless flow, from film feeding to the final sterilized product?
A missing machine part stops your whole production line. You lose time and money when machines do not connect well. You need a complete and perfect system.
Essential machinery includes film feeding units, bag forming stations, precise filling nozzles, sealing units, and terminal sterilizers. Conveyor belts must connect these parts smoothly to keep the product moving without human touch.
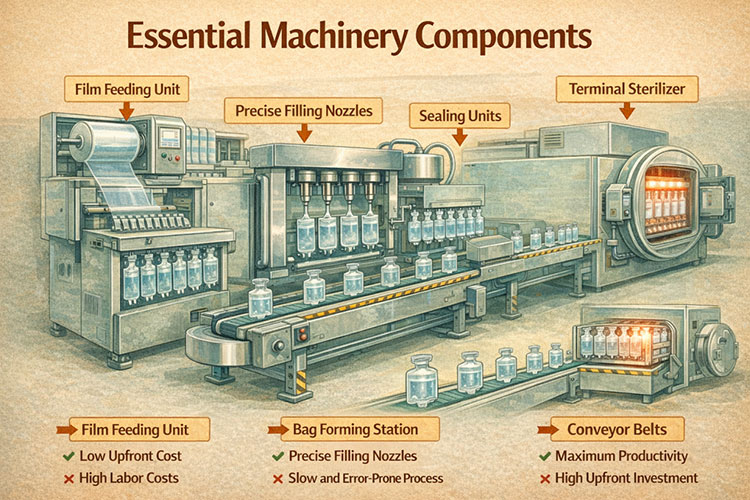
Essential Machinery Components
The Core Machines
Every machine part must work together perfectly. I compare it to a car engine. The car stops if one part breaks. The film feeding unit is the start of the line. It holds the big rolls of plastic. It must feed the plastic smoothly. The bag will leak if the plastic wrinkles. Next is the forming station. This part uses strong heat to shape the bag. Then we have the filling nozzles. They put the liquid drug into the bag very quickly.
After that, the sealing unit closes the bag tightly. Finally, the bags go into the terminal sterilizer. This is a big steel oven. It uses hot steam to kill all bacteria. You need strong conveyor belts to move the bags between these machines. We use stainless steel for all parts. Stainless steel does not rust easily. It is very easy to clean. We test every connection before we ship the line.
| Machine Component | Main Function |
| Film Feeding Unit | Holds and unrolls raw plastic material |
| Forming Station | Shapes the plastic into a bag |
| Filling Nozzles | Injects the liquid drug into the bag |
| Sealing Unit | Closes the bag with high heat |
| Terminal Sterilizer | Kills all bacteria with hot steam |
5.How does a high-speed linkage mode (up to 450 bpm for glass or 7500 bags/h for soft bags) impact the overall efficiency of an IV fluid factory?
Slow machines limit your daily output. You miss big orders if your line cannot keep up. High-speed linkage solves this problem completely.
High-speed linkage connects all machines to work at the same fast pace. Running at 7500 bags per hour lowers your cost per unit, cuts down labor time, and massively boosts your factory output.

High Speed Linkage Mode
Speed and Factory Output
Speed is money in the pharmaceutical business. I visited a factory last year. Their old machines ran very slowly. They could not accept big orders from the government. We installed a new high-speed linkage line for them. The new line makes 7500 soft bags every hour. This change saved their business completely. High-speed linkage means all machines talk to each other. The film feeding machine slows down if the filling machine slows down.
They work as one big team. This communication stops traffic jams on the conveyor belt. Your cost for each bottle drops when you make 450 glass bottles per minute. You use the same amount of electricity and space. But you make more products to sell. You can sell your IV fluids at a better price. You beat your competitors easily. We use smart servo motors to keep the fast speed very stable.
| Speed Mode | Daily Output (8 hours) | Cost Per Unit |
| Low Speed (Old Tech) | 10,000 bags | High |
| Medium Speed | 30,000 bags | Medium |
| High-Speed Linkage | 60,000+ bags | Very Low |
6.What is the critical role of filling accuracy (±0.3%) in maintaining high ROI and meeting strict pharmacopeia standards?
Overfilling wastes expensive liquid drugs. Underfilling causes batch rejections and audit failures. Poor accuracy ruins your profit margin very fast.
A strict filling accuracy of ±0.3% prevents product waste and ensures patient safety. This precision helps you pass strict pharmacopeia standards and protects your Return on Investment (ROI) by saving valuable liquid drugs.

Filling Accuracy and Precision
The Value of Precision
Liquid drugs are very expensive to make. I always tell my clients to protect their liquid. You lose money if your filling machine puts too much liquid in the bag. Imagine losing one milliliter in every single bag. You lose a lot of expensive medicine if you make 100,000 bags a day. You have a bigger problem if you put too little liquid. The hospital will test the bag. They will see the volume is wrong.
They will reject the whole batch. The government might close your factory. We use advanced mass flow meters. We also use high-quality ceramic piston pumps. These tools give us a filling accuracy of ±0.3%. This number means every bag is almost perfect. You pass the strict pharmacopeia standards easily. You do not waste medicine. You keep your profit high. Precision is the most important part of liquid filling.
| Filling Error | Result | Impact on Business |
| Overfilling | Wastes liquid drug | Lowers profit margin |
| Underfilling | Fails hospital tests | Causes batch recall and bad reputation |
| ±0.3% Accuracy | Perfect volume | Passes audits and saves money |
7.How do we integrate purified water systems (RO+EDI) and distilled water generators to ensure the continuous supply of high-purity WFI (Water for Injection)?
Bad water quality ruins your IV fluids. Your whole factory stops if your water system fails. You must integrate a safe and strong water system.
We connect Reverse Osmosis (RO) and Electrodeionization (EDI) systems to multi-effect distilled water generators. This setup continuously filters and heats the water, ensuring a steady and safe supply of Water for Injection (WFI).

Purified Water Systems RO EDI
Making Safe Water
Water is the main ingredient in IV fluids. You cannot use normal city water. Normal water has minerals and bacteria. I have seen bad water ruin a whole month of hard work. You must build a perfect water system. First, we use a Reverse Osmosis (RO) system. This machine forces water through a very tight filter. It removes most bad things. Then we use an Electrodeionization (EDI) unit. This part uses electricity to remove the last minerals.
Now you have purified water. But IV fluids need Water for Injection (WFI). WFI is the cleanest water in the world. We send the purified water into a multi-effect distilled water generator. This big machine boils the water into steam. It does this many times. The steam turns back into water. This water is totally sterile. We store it in a hot tank. We keep it moving in the pipes so bacteria cannot grow.
| Water System Step | Technology Used | Purpose |
| Step 1 | Reverse Osmosis (RO) | Removes large particles and most minerals |
| Step 2 | Electrodeionization (EDI) | Removes remaining trace minerals |
| Step 3 | Distilled Water Generator | Boils water to create sterile WFI |
| Step 4 | Hot Loop Storage | Keeps WFI moving to stop bacteria growth |
8.What are the challenges in designing HVAC and BMS systems to precisely control temperature, humidity, and pressure difference in a Class A/B cleanroom?
Poor air control brings bacteria into your cleanroom. This mistake ruins your sterile products and causes huge recalls. HVAC design is very hard.
The main challenge is keeping exact pressure differences between rooms to stop dirty air from entering. The HVAC and Building Management System (BMS) must constantly adjust airflow to maintain Class A/B cleanroom standards.

HVAC and BMS Cleanroom Control
Controlling the Air
You cannot see the air. But the air can destroy your business. A cleanroom is not just a clean room. It is a highly controlled box. I help clients understand the HVAC system. HVAC stands for Heating, Ventilation, and Air Conditioning. It pushes clean air through HEPA filters. These thick filters catch tiny dust and bacteria. The Building Management System (BMS) is the brain. It watches the room all day and night.
The biggest challenge is air pressure. You must keep the clean room at a higher pressure than the dirty room. The clean air pushes out when you open a door. The dirty air cannot come in. The BMS sounds a loud alarm if the pressure drops. We design the air ducts very carefully. We make sure the temperature stays the same everywhere. We make sure the humidity stays the same. This control keeps your liquid drugs safe and stable.
| Cleanroom Factor | Control Method | Why it matters |
| Air Particles | HEPA Filters | Stops bacteria from touching the drug |
| Pressure Difference | HVAC Fan Speed | Pushes dirty air away from clean areas |
| Temperature | Cooling/Heating Coils | Keeps the liquid drug stable |
| Humidity | Dehumidifiers | Stops mold growth in the room |
9.How does a centralized CIP (Cleaning-in-Place) system simplify the maintenance and cross-contamination prevention of the entire production line?
Taking machines apart to clean them wastes many hours. Manual cleaning also leaves dirt behind. This dirt causes cross-contamination between batches.
A centralized CIP system pumps cleaning fluids and hot steam through the pipes and tanks automatically. It cleans the machines without taking them apart, saving time and completely preventing cross-contamination between different drug batches.

Cleaning in Place CIP System
Easy and Safe Cleaning
Cleaning machines by hand is a terrible job. I remember a factory where workers spent six hours taking pipes apart to clean them. They lost a whole day of production. Sometimes they forgot to clean a small corner. The next batch of drugs got contaminated. A centralized CIP system changes everything. CIP stands for Cleaning-in-Place. You do not take the machines apart anymore. You just press a button on the computer screen.
The system pumps special acid and base liquids through all the pipes. It washes the inside of the big filling tanks. Then it pumps clean water to rinse everything. After that, we use SIP (Sterilization-in-Place). The system pushes very hot pure steam through the pipes. The hot steam kills every single germ. This process is very fast. It is perfectly safe. You can switch from making one drug to another drug quickly. You never worry about cross-contamination again.
| Cleaning Method | Time Needed | Risk of Contamination |
| Manual Cleaning | 6 to 8 hours | High (human error) |
| CIP/SIP System | 1 to 2 hours | Zero (automated and sterile) |
10.What does a "Seamless Customer Experience" look like during the transition from equipment manufacturing to onsite installation?
Getting big machines delivered is stressful. Your factory launch gets delayed if the installation team is bad. You need a smooth handover process.
A seamless experience means we build the machines, test them in our factory, and then send our engineers to your site. We handle the setup, connect the utilities, and train your workers to run the line.

Equipment Installation and Training
From Factory to Your Site
Buying a big production line is scary. You send a lot of money to another country. You worry if the machines will actually work. I understand this deep fear. We create a seamless customer experience to take away your stress. First, we build the machines in our factory in China. Then we invite you to come visit. We do a Factory Acceptance Test (FAT). We run the machines with your bags and your bottles.
You check everything carefully. We pack the machines safely if you are happy. We ship them to your country. But our job is not done. We send our best engineers to your factory. They unpack the machines. They connect the electricity and the water pipes. They do a Site Acceptance Test (SAT). They teach your workers how to press the buttons. They teach them how to fix small problems. We stay until your first real batch is perfect.
| Project Stage | Action | Customer Benefit |
| Manufacturing | Build machines to your exact design | Get exactly what you ordered |
| FAT | Test machines in our factory | See proof that the machine works |
| Installation | Our engineers set up the line | No stress about assembly |
| SAT & Training | Test on site and teach workers | Fast and smooth factory launch |
11.How do we navigate the complexities of GMP, FDA, and WHO validation (DQ, IQ, OQ, PQ) to ensure the factory is legally ready for production?
Failing a validation test keeps your factory closed. You cannot sell drugs without GMP or FDA approval. The paperwork is completely overwhelming.
We provide full validation documents, including Design, Installation, Operational, and Performance Qualifications (DQ, IQ, OQ, PQ). We guide you through the testing process to prove your factory meets GMP, FDA, and WHO rules.
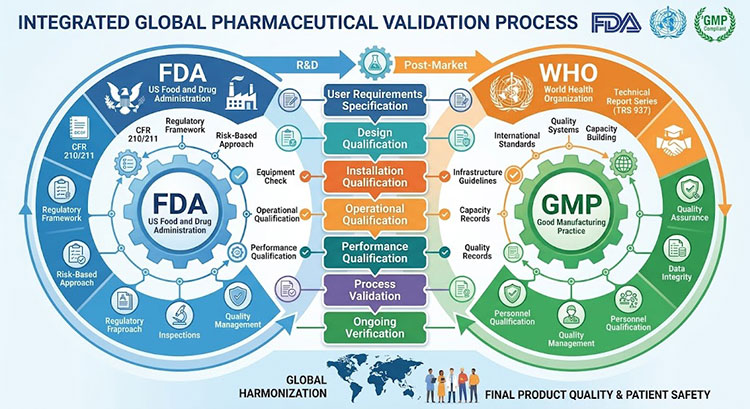
GMP FDA WHO Validation Process
Passing the Government Tests
You cannot sell medicine just because your machines run. You must prove to the government that your factory is safe. The FDA and WHO have very strict rules. Good Manufacturing Practice (GMP) is the law. I help our clients with the hard paperwork. We give you a huge book of documents. This process is called validation. First is Design Qualification (DQ). We prove the paper design is correct.
Second is Installation Qualification (IQ). We prove we installed the machines exactly like the design. Third is Operational Qualification (OQ). We turn the machines on. We prove they run at the right speed and temperature. Last is Performance Qualification (PQ). We run the machines with real products for many days. We test the final bags in a laboratory. We prove the whole system makes perfect medicine every time. You show these papers to the inspector. You pass the audit easily.
| Validation Step | Full Name | What it proves |
| DQ | Design Qualification | The engineering plan meets GMP rules |
| IQ | Installation Qualification | The machines are built and placed correctly |
| OQ | Operational Qualification | The machines run at the right settings |
| PQ | Performance Qualification | he whole system makes safe drugs constantly |
12.How does AIPAK’s experience with over 800 projects help in anticipating and solving tricky engineering problems before they cause downtime?
Inexperienced suppliers make mistakes that stop your line. You pay the price for their learning curve. Deep experience is your best protection.
With over 15 years and 800 projects, we have seen every possible layout issue and machine fault. We use this deep experience to spot problems early, fix them in the design phase, and prevent future downtime.

AIPAK Engineering Experience
Learning from Real Projects
Experience is the most valuable thing in engineering. Books only teach a little. Our company has finished more than 800 projects over the last 15 years. I have traveled to many different countries. I have seen factories in hot deserts and cold mountains. Every environment brings different problems. Once, a client in a very humid city had water dripping from their cleanroom ceiling. We knew how to fix their air system quickly. We saw this in another country before.
We use our past mistakes to make your design perfect. We know which machine parts wear out the fastest. We give you extra spare parts before you even ask. We know how to arrange the machines. Your workers do not get tired. You do not just buy steel and wires when you choose us. You buy 15 years of deep knowledge. You buy total peace of mind.
| AIPAK Advantage | How We Do It | Benefit to You |
| 800+ Projects | We use past data for new designs | Zero beginner mistakes in your factory |
| Global Experience | We adjust designs for local weather | Machines run perfectly in any climate |
| Spare Parts Planning | We predict which parts will break | No long waiting times for repairs |
Conclusion
Building an IV fluid line requires perfect design and reliable machines. We provide the complete solution to help you succeed in safe, compliant, and efficient pharmaceutical manufacturing.
Don't forget to share this post!
CONTACT US
Tell us your raw material and project budget to get quotations within 24 hours.
WhatsApp Us: +86 181 7101 8586
 Tell us your material or budget, we'll reply you ASAP within 24 hours
Tell us your material or budget, we'll reply you ASAP within 24 hours

