How Blow-Fill-Seal (BFS) Technology Redfines Contamination Control in Pharma Packaging?
Product recalls ruin brands. Sterile liquid contamination puts patients at serious risk. Blow-Fill-Seal (BFS) technology steps in to stop these dangerous errors at the source.
Blow-Fill-Seal (BFS) technology is an automated manufacturing process that forms, fills, and seals a plastic container in one continuous operation. This closed system removes human contact, blocks outside air, and creates a sterile environment to redefine contamination control in pharma packaging.

Blow-Fill-Seal technology pharma packaging
You might think your current cleanroom setup is safe enough. But hidden risks still exist on your production floor. If you want to protect your products and your patients, you need to see how this technology changes everything.
1.What Is Blow-Fill-Seal Technology and How Does It Work?
Buying empty vials and filling them later takes too much time. It also adds contamination risks. BFS technology solves this by doing everything inside one machine.
BFS technology works by melting plastic resin into a tube. The machine blows the tube into a mold to form a container. It then fills the container with liquid and seals it immediately. This all happens in seconds inside a sterile machine zone.
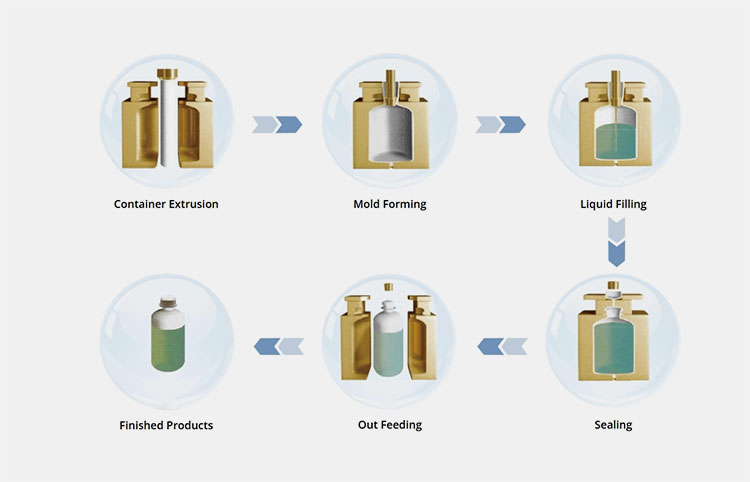
How Blow-Fill-Seal works
I remember visiting a client's factory five years ago. They struggled with a slow glass vial filling line. Workers had to wash, sterilize, fill, and seal the vials in different steps. Each step added a chance for bacteria to enter. I told them about Blow-Fill-Seal machines. This process changes the whole game.
The Three Main Steps of BFS
The BFS process relies on three basic actions. First, the machine heats plastic pellets until they melt. This high heat kills any bacteria on the plastic. Second, the machine shapes the hot plastic into a bottle or ampoule. Third, special nozzles fill the medicine into the plastic shape, and the machine seals the top right away.
We can look at the differences between old methods and BFS in this table.
| Process Step | Traditional Glass Filling | Blow-Fill-Seal (BFS) |
| Container Prep | Wash and bake glass vials | Melt plastic resin |
| Sterilization | Needs separate large ovens | Heat melts and sterilizes |
| Filling | Open to cleanroom air | Closed inside the mold |
| Sealing | Add rubber stopper and cap | Melt plastic to close top |
| Time Taken | Hours to complete | Seconds to complete |
This simple table shows why BFS is faster and safer. The entire cycle takes less than 15 seconds. The medicine never touches the outside air. This fast action stops dust and germs from ruining the batch.
2.Why Is Contamination Control So Critical in Pharma Packaging?
A tiny speck of dust can ruin a whole batch of medicine. Recalls cost millions of dollars and destroy trust. Strict contamination control prevents these costly disasters.
Contamination control is critical because sterile medicines go directly into the human body, like eye drops or injections. Any bacteria or particles in the liquid can cause severe infections. Good packaging blocks germs, keeps patients safe, and meets strict FDA rules.

Contamination control in pharma
In my 15 years working with pharma packaging equipment, I have seen companies lose big money due to poor cleanroom rules. One client had to throw away 50,000 bottles of eye drops. A worker sneezed near an open filling line. The filters did not catch all the germs. This is why contamination control matters so much.
The Hidden Dangers in Open Filling
Every time a container stays open, danger grows. Dust floats in the air. People shed skin cells. Machines create tiny metal flakes. If any of these things fall into a medicine bottle, the product becomes poison.
Here is a breakdown of common contamination sources:
| Contamination Source | How It Enters | Risk Level |
| Human Operators | Touching parts, breathing, shedding hair | Very High |
| Cleanroom Air | Poor airflow, bad filters | High |
| Packaging Materials | Dirty empty glass bottles or caps | High |
| Machine Parts | Oil leaks, metal dust from moving parts | Medium |
We must control all these sources. Sterile packaging is not just a nice idea. It is the law. Government agencies like the FDA demand proof that your medicine is clean. If you fail their tests, they will shut down your factory. BFS technology gives you the best defense against these strict rules.
3.How Does BFS Technology Eliminate Human Intervention Risks?
People make mistakes. Even well-trained workers carry germs into cleanrooms. BFS technology removes the human element from the core filling process to ensure pure safety.
BFS technology eliminates human intervention by enclosing the entire form, fill, and seal process inside a sealed machine chamber. Workers do not load empty bottles or apply caps. They only monitor the machine from the outside through computer screens.

BFS machine automation
I often tell my clients that humans are the dirtiest things inside a cleanroom. Even with special suits, masks, and gloves, a person still releases thousands of particles every minute. I once watched an operator adjust a filling nozzle by hand. That small touch ruined the sterility of the whole line.
Replacing Hands with Automated Systems
BFS machines fix this human problem. The machine takes raw plastic pellets from a closed pipe. It does the rest of the work inside a Class A air shower area. No human hands ever reach into this critical zone while the machine runs.
Let us compare human actions versus BFS automation.
| Task | Human Operator Action | BFS Machine Action |
| Moving empty bottles | Carry boxes, load onto belts | Machine makes bottles inside |
| Placing caps | Sort caps, fix jammed caps | Machine melts plastic to seal |
| Fixing errors | Open doors, reach inside | Sensors auto-reject bad items |
| Cleaning | Wipe surfaces with chemicals | Clean-In-Place (CIP) systems |
Because the machine does everything, you need fewer people in the room. Fewer people mean fewer germs. The BFS system uses strong air flows to blow any loose particles away from the open plastic tubes. This smart design keeps the product perfectly safe without needing human help.
4.What Are the Core Advantages of BFS Over Traditional Glass Vials?
Glass vials break easily during shipping. Broken glass causes product loss and safety hazards. BFS plastic containers offer a stronger, safer, and cheaper alternative.
The core advantages of BFS over glass include a lower risk of breakage, lighter shipping weight, and a simplified supply chain. BFS uses medical-grade plastic that does not shatter. It also removes the need to buy, wash, and store large amounts of empty glass containers.

BFS plastic vs glass vials
Many older pharma factories still love glass. But I always ask them about their breakage rates. One client told me they lose about 2% of their product just from glass breaking on the line. That sounds small, but it equals thousands of dollars every month. I helped them look at BFS as a better option.
5.Why Plastic Wins in Modern Packaging?
Glass is heavy and fragile. It also needs rubber stoppers and aluminum seals. These extra parts complicate the supply chain. You have to buy from three different suppliers. With BFS, you only buy plastic resin.
Here is a clear comparison between the two materials.
| Feature | Traditional Glass Vials | BFS Plastic Containers |
| Durability | Breaks easily if dropped | Squeezable and shatterproof |
| Weight | Heavy, costs more to ship | Very light, cheap to ship |
| Supply Chain | Buy glass, rubber, and metal | Buy only plastic pellets |
| Storage Space | Needs huge warehouses for empty glass | Needs small space for resin bags |
| Patient Safety | Risk of glass shards in liquid | No glass shard risk |
Patients also prefer plastic. If a nurse drops a BFS ampoule on the floor, it bounces. If they drop a glass vial, it shatters into dangerous pieces. BFS makes the final product much easier to handle in hospitals and homes.
6.How Does BFS Fit Into Modern GMP and Sterile Packaging Trends?
Old machines struggle to pass new FDA rules. Upgrading your factory is hard and stressful. BFS technology fits perfectly into modern Good Manufacturing Practices (GMP) to secure your future.
BFS fits into modern GMP trends by providing built-in data tracking, automated cleaning, and strict environmental control. It aligns with the global push for smaller cleanrooms and less human contact. Regulatory bodies strongly favor BFS for its reliable and repeatable sterile results.
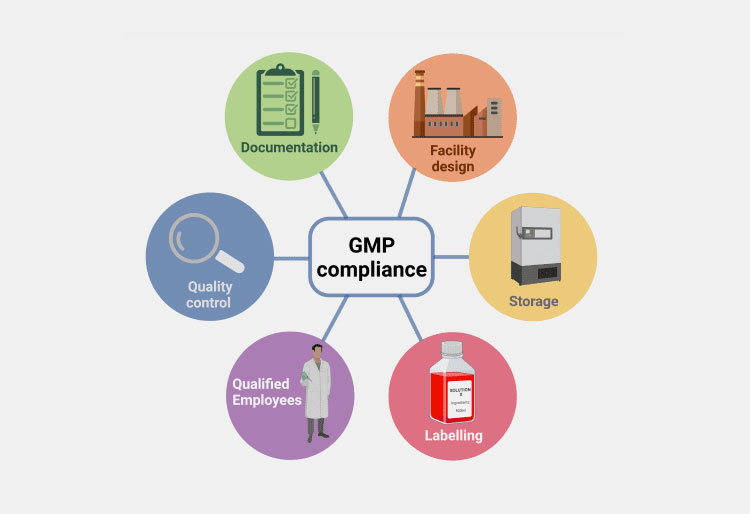
BFS and GMP compliance
In our 15 years at AIPAK ENGINEERING, we have seen GMP rules get stricter every year. I used to help clients pass inspections with basic filling lines. Now, inspectors want to see advanced automation and data logs. They want proof that every single bottle is safe. BFS systems provide this proof easily.
The Future of Sterile Manufacturing
Modern factories want to shrink their cleanrooms. Big cleanrooms cost too much money to run. Because a BFS machine creates its own sterile zone inside, you can put it in a less strict room background. This saves massive amounts of energy.
Look at how BFS matches current industry trends.
| Industry Trend | How BFS Supports It |
| Smaller Cleanrooms | BFS acts as its own mini-cleanroom |
| Data Integrity | Machines record every temperature and pressure |
| Less Human Contact | Fully automated from raw plastic to sealed bottle |
| Green Manufacturing | Plastic is lightweight and highly recyclable |
| Rapid Production | Makes thousands of units per hour without stopping |
The future of pharma packaging is automation and closed systems. We focus on helping our B2B clients upgrade to these smart solutions. When you adopt BFS, you do not just buy a machine. You buy peace of mind and long-term GMP compliance.
Conclusion
Choosing BFS technology is a strategic investment in absolute sterility and operational efficiency. By eliminating human intervention and streamlining production into a single automated step, you secure your brand's reputation and significantly boost your factory's ROI. Ready to upgrade your production capabilities? Contact AIPAK ENGINEERING’s engineering team today for a customized BFS technical proposal and cleanroom layout design tailored to your specific pharmaceutical needs
Don't forget to share this post!
CONTACT US
Tell us your raw material and project budget to get quotations within 24 hours.
WhatsApp Us: +86 181 7101 8586
 Tell us your material or budget, we'll reply you ASAP within 24 hours
Tell us your material or budget, we'll reply you ASAP within 24 hours

